Executive Summary
Ozonation (also referred to as ozonisation) is a chemical water treatment technique based on the infusion of ozone into water. Ozone is a gas composed of three oxygen atoms (O3), which is one of the most powerful oxidants. Ozonation is a type of advanced oxidation process, involving the production of very reactive oxygen species able to attack a wide range of organic compounds and all microorganisms. The treatment of water with ozone has a wide range of applications, as it is efficient for disinfection as well as for the degradation of organic and inorganic pollutants. Ozone is produced with the use of energy by subjecting oxygen (O2) to high electric voltage or to UV radiation. The required amounts of ozone can be produced at the point of use but the production requires a lot of energy and is therefore costly.
| In | Out |
|---|---|
Freshwater, Drinking Water, Blackwater, Greywater, Non-biodegradable Wastewater, Treated Water, Energy |
Drinking Water, Treated Water |
Introduction
Ozone (O3) has been used in water treatment since the late 19th century. Today it is applied for the disinfection of drinking water, for the removal of effluents from wastewater treatment plants in a process called ozonation (or ozonisation) as well as for the degradation of organic and inorganic pollutants in wastewater (see also advanced oxidation process).
Disinfection with ozone
Ozone is an excellent disinfectant and can even be used to inactivate microorganisms such as protozoa, which are very resistant to conventional disinfectants (VON GUNTEN 2003). However, ozone is an unstable gas that transforms to oxygen hence no residual disinfection effect takes place with ozonation.
Degradation of organic pollutants with ozone
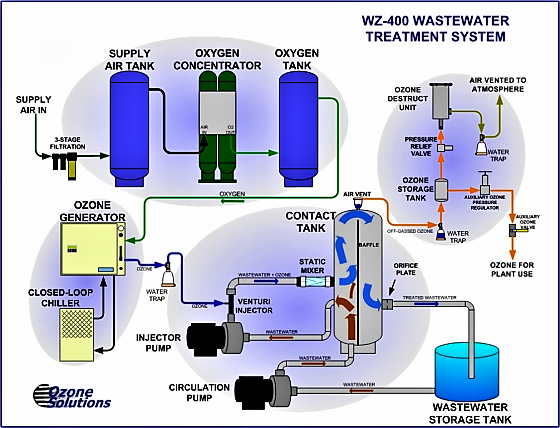
Ozonation is an efficient treatment to reduce the amounts of micropollutants released in the aquatic systems by wastewater treatment plants (MARGOT et al. 2011). Although no residual by-products are generated by ozone itself, some concerns are raised regarding oxidation by-products when water containing both organics and ions, such as bromide, iodide and chlorine ions, are treated with ozonation. A typical ozonation system consists of an ozone generator and a reactor where ozone is bubbled into the water to be treated.
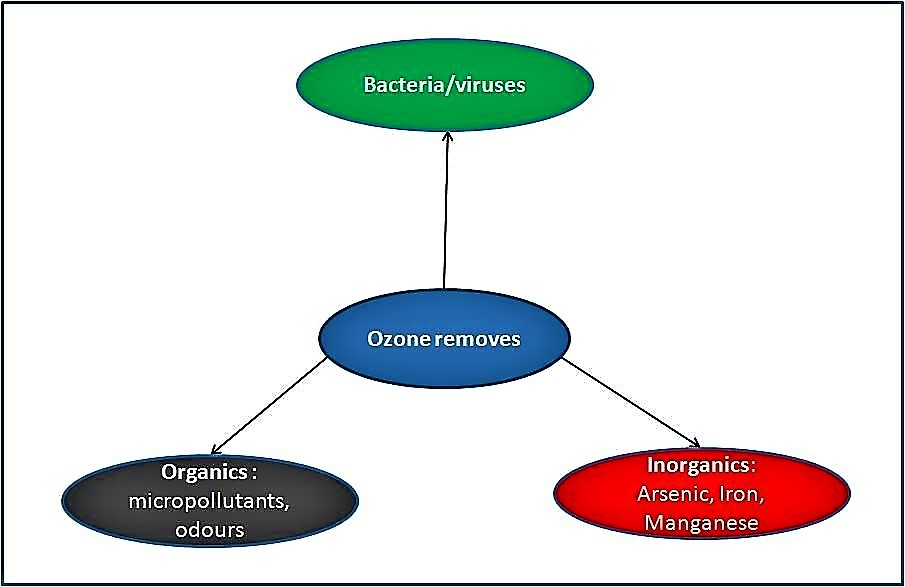
The effect of ozone
The effectiveness of ozone results from its powerful oxidising effect on chemicals and microorganisms caused by the generation of reactive oxygen species during ozone transformation to oxygen. Ozone directly attacks the surface of microorganisms and destroys their cell walls. The cells thus loose their cytoplasm and can no longer reactivate themselves (STUCKI et al. 2005). Ozone can induce an oxidative degradation of many organics and leaves more biodegradable compounds (DERCO et al. 2001). Besides, ozone can oxidise metallic ions such as Fe(II), Mn(II) or As(III) producing insoluble solid oxides that can be easily separated from water by filtration or sedimentation.
Production of ozone
Because of its relatively short half-life, ozone is generated on-site by an ozone generator. The conventional ways to produce ozone are UV-light and corona-discharge. Ozone generation by corona-discharge is most common nowadays and has many advantages such as longer lifespan of the unit, higher ozone production and higher cost efficiency. Production with UV-light is an option where only small amounts of ozone are required. Other ozone generators that are available involve electrolysis of water and the use of membranes. With this method, the ozone is dissolved in the process water as soon as it is formed resulting in ozonation using minimum equipment.
Technical aspects relative to disinfection
Adapted from WHO (2009)
Whilst ozone is the most effective disinfectant overall and is more effective than chlorine in inactivating viral agents, there are significant disadvantages to its use. Ozone does not provide residual protection against recontamination during distribution and as ozone affects biological stability, it may encourage re-growth of bacteria. However, given the concerns about the use of chlorine in many countries because of the formation of toxic disinfection by-products (DBP), the use of ozone is increasingly investigated and the lack of residual may be dealt with by employing regular booster ozonation during distribution.
Cost considerations
The cost of ozone disinfection systems depends on the manufacturer, the site, the capacity of the plant, and the characteristics of the wastewater to be disinfected. Ozonation costs are generally high in comparison to other disinfection techniques (EPA 1999). For the removal of micropollutants in wastewater, the additional operation costs for ozonation combined with sand filtration are around 3-4 Swiss cents/m3 (MARGOT et al. 2011).
Operation and maintenance
Adapted from EPA (1999)
Ozone generation uses a significant amount of electrical power. Constant attention must thus be given to the system to ensure that power is available. Moreover, ozone should not be released from the system and connections in or surrounding the ozone generator should not be leaking. The operator must monitor the appropriate subunits on a regular basis to ensure that they are not overheated. Therefore, the operator must check for leaks routinely since a very small leak can cause unacceptable ambient ozone concentrations.
Health aspects
There are significant health and safety concerns for operators regarding the production and application of ozone. However, far less is known about ozonation and the effect of ozone on human health (WHO 2009). There are also concerns regarding by-product formation during the disinfection of drinking water containing bromide ions (VON GUNTEN 2003).
At a glance
| Working principle | Infusion of ozone, a gas produced by subjecting oxygen molecules to high electrical voltage, which reacts with microorganisms and pollutants |
| Capacity/adequacy | High tech equipment required |
| Performance | High efficiency |
| Costs | Relatively high operation costs |
| Self-help compatibility | Engineers are required for the design |
| O&M | Continuous input of electrical power required |
| Reliability | Reliable if operating conditions are scaled taking into account wastewater content |
| Main strength | Very efficient and fast method for disinfection and as a AOP |
| Main weakness | Requires complicated equipment as well as large amounts of energy and qualified operators |
Ozonation has been successfully applied for water disinfection and can kill most bacteria viruses and protozoa. However, there is no residual disinfection effect and ozonation is more expensive than chlorination. Ozonation is a suitable process to degrade organic pollutants (e.g. for micropollutants removal and landfill leachate pre-treatment) and oxidise metallic ions (e.g. iron manganese).
Design and construction needs skilled staff and high-tech equipment. Sophisticated generators consuming high-amounts of electricity are required to produce ozone. Although operation and maintenance costs are relatively low, precise monitoring and dosing adjustment of ozone is needed to ensure the efficiency of the treatment (NABI BIDHENDI et al. 2006).
Influence of Ozonation on Biodegradability of Refractory Organics in a Landfill Leachate
Wastewater Technology Fact Sheet Ozone Disinfection
Ozonation of Drinking Water: Part II. Disinfection and By-products Formation in the Presence of Bromide, Iodide or Chlorine
Elimination of Micropollutants in wastewater Treatment Plants: Ozonation or Activated Carbon? Conclusions of a One-year Pilot Project
Application of Ozonation in Drinking Water Disinfection Based on an Environmental Management Strategy Approach Using SWOT Analysis
Ozonization of Purified Water Systems
Scheme of a typical ozonation process
WHO Seminar Pack for Drinking Water Quality
Ozone in Drinking Water Treatment a Brief Overview 106 Years and Still Going
This is a review article about ozonation in drinking water including many aspects such as ozone production, reaction mechanisms, applicability, price, safety, etc.
EAGLETON, J. (1999): Ozone in Drinking Water Treatment a Brief Overview 106 Years and Still Going. URL [Accessed: 23.05.2019]Modelling of Micropollutant Removal by Ozonation and Chlorination in Potable Water Treatment
This report describes the building up of a new simulator for chlorination and ozonation. It aims at giving a clear overview of the investigation fields related to modeling of oxidation for water purification. The model proposed is adaptable to on-site conditions.
MANDEL, P. (2007): Modelling of Micropollutant Removal by Ozonation and Chlorination in Potable Water Treatment. In: TECNEAU FP: Volume 6 URL [Accessed: 23.05.2019]Advanced Treatment by Ozonation and Sonolysis for Domestic Wastewater Reuse
This publication describes the combination of ozone and ultrasounds for the disinfection and discoloration of domestic wastewater treatment plant effluents for reuse purposes.
SELCUK, H. ; NADDEO, V. ; BELGIONO, V. (2007): Advanced Treatment by Ozonation and Sonolysis for Domestic Wastewater Reuse. In: Proceedings of the 10th International Conference on Environmental Science and Technology: URL [Accessed: 21.05.2019]Experiences During 25 Years of Ozonation for Different Industrial Applications
This article provides case studies on different industrial applications of ozonation.
STOPKA, O. (1997): Experiences During 25 Years of Ozonation for Different Industrial Applications. In: IOA/PAG Conference: URL [Accessed: 03.10.2011]Evaluating the Use of Ozone for Disinfection of Drinking Water, Case Study: Tehran Pars Water Treatment Plant (Iran)
In this study, the use of ozone was taken into consideration for disinfection of drinking water in Tehran. Advantages and disadvantages of water ozonation are evaluated with a pilot plant in Tehran Pars water treatment plant.
HOVEID, H. ; NABI BIDHENDI, G. ; REZA JAFARI, H. ; NASRABADI, T. ; SHAHRIARI, T. (2008): Evaluating the Use of Ozone for Disinfection of Drinking Water, Case Study: Tehran Pars Water Treatment Plant (Iran) . In: Environmental Sciences : Volume 5 , 31-38. URL [Accessed: 03.10.2011]Traitement des Micropolluants dans les Eaux Uses-Rapport Final sur les Essais Pilotes a la STEP de Vidy (Lausanne)
This document presents the results of a pilot study aiming at removing water micropollutants at the Lausanne (Switzerland) wastewater treatment plant.
MARGOT, J. ; MAGNET, A. ; THONNEY, D. ; CHEVRE, N. ; ALENCASTRO, F. de ; ROSSI, L. (2011): Traitement des Micropolluants dans les Eaux Uses-Rapport Final sur les Essais Pilotes a la STEP de Vidy (Lausanne). In: Ed. Ville de Lausanne : URL [Accessed: 21.05.2019]Ozonation of Cooling Tower Water: A Case Study
This report provides background information on the use and treatment with ozonation of recirculating cooling water systems. Details about the technology employed at the study site, the water savings, other results, and the costs and savings are discussed. The report identifies factors that should be taken into account when ozone is considered for cooling tower water treatment.
OSGOOD, S. (1991): Ozonation of Cooling Tower Water: A Case Study . In: California Department of Water Resource. [Accessed: 29.01.2012].Ozone Planet
This study describes the ozonation facilities installed in Yorkshire for pesticide removal and algae control to enhance the slow sand filters without the need for granular activated carbon.
OZONIA (2000): Ozone Planet. Loftsome Bridge Yorkshire Water Services. [Accessed: 03.10.2011] PDFOzone Water Treatment Application and Design
Presentation on different ozone generators, processes design and monitoring.
SACCO, A. (2009): Ozone Water Treatment Application and Design. In: Spartan Environmental Technologies LLC.: URL [Accessed: 23.05.2019]Combined Ozonation-Nanofiltration for Drinking Water Treatment
This presentation shows the benefits of combining ozone with nanofiltration for water treatment namely the reduction of membranes fouling.
KARNIK, B.S. ; CHEN, K.C. ; JAGLOWSKI, D.R. ; DAVIES S.H. ; BAUMANN, M.J. ; MASTEN S.J. (2004): Combined Ozonation-Nanofiltration for Drinking Water Treatment. In: United State Environmental Protection Agency: URL [Accessed: 23.05.2019]Water Treatment Technology Factsheet on Ozone
This short factsheet describes the process, technical limitations and capabilities as well as the cost of ozonation for water treatment.
ALLCONSULTING (n.y): Water Treatment Technology Factsheet on Ozone . In: Water Treatment Catalog and Decision Tool: URL [Accessed: 03.10.2011]ozone.ch - your specialist for water treatment and ozone !
This website provides information on water treatment studies, computing, equipment, and components related to ozonation.
Drinking Water Treatment Methods
Summaries of different water treatment methods including ozonation are presented in this website.